eLabels as a Patient Access Hub: Exploring Extensibility Across the Clinical Trial Ecosystem
Electronic labels (eLabels) are often positioned as a digital replacement for paper labels as they improving accessibility, version control, and regulatory compliance. While these benefits are real and important, they represent only the baseline value of eLabels.
The more transformative opportunity lies in extensibility: using eLabels as a trusted, always-available digital access point that connects patients to the broader set of systems, data, and information they rely on throughout a clinical trial.
From a patient’s perspective, participation in a trial extends well beyond taking study medication. Patients must navigate instructions, schedules, consent materials, reporting tools, and multiple points of contact which are often spread across disconnected systems. eLabels, accessed through a simple QR code are uniquely positioned to act as a unifying gateway into this ecosystem.
Study and Treatment Information
At its core, a clinical trial asks patients to understand what they are taking and why it matters. Clear, accessible study and treatment information helps patients feel confident about how to take their medication, how to store it, and what to expect along the way. When this information is easy to find and written in plain language, it reduces anxiety and removes guesswork from day-to-day participation. eLabels provide a natural, familiar place for this information to live and stay current as the study evolves.
Instructions and Reference Materials
Beyond the label itself, patients often need additional guidance to use a medication or device correctly. This may include instructions for use, quick reference guides, or short “how-to” videos that reinforce what they were shown at the site. When these materials are easy to access at the moment they are needed, patients are more likely to follow instructions correctly and consistently. Linking these resources through an eLabel keeps everything in one trusted place, even when the content itself is hosted elsewhere.
Study Participation Tools
Many clinical trials now ask patients to interact with digital tools as part of their participation, such as symptom trackers or daily eDiaries. These tools are essential, but they can feel fragmented when access depends on separate apps, emails, or login details. Providing a simple link from the eLabel allows patients to move seamlessly from taking their medication to recording their experience. This connection makes participation feel more intuitive and less like a series of disconnected tasks.
Consent and Regulatory Documentation
Informed consent is not a one-time event, but an ongoing part of a patient’s relationship with a clinical trial. Patients may want to revisit what they agreed to, review updates, or better understand changes introduced during the study. Easy access to consent and regulatory documentation supports transparency and reinforces trust between the patient, the site, and the sponsor. Linking this information through an eLabel reduces administrative friction while helping patients stay informed throughout the study.
Visit Planning and Logistics
Clinical trial participation often requires careful coordination of visits, appointments, and preparation steps. Patients benefit when they can easily see what is coming next, when they are expected on site, and how to prepare for upcoming visits. When sites use electronic scheduling systems, a link from the eLabel can remove uncertainty and reduce missed or misunderstood visits. Emails are easily lost and portals easily forgotten, but a patient always has their medication, making the eLabel a reliable way to find what they need, when they need it.
Support and Escalation Channels
At some point, most patients will have a question, concern, or need for reassurance during a trial. Knowing exactly who to contact and how to reach them can make the difference between a minor issue and a larger problem. When support and escalation information is accessible directly from the eLabel, patients are not dependent on paperwork or old messages. Even if everything else is misplaced, their medication provides a direct path to help.
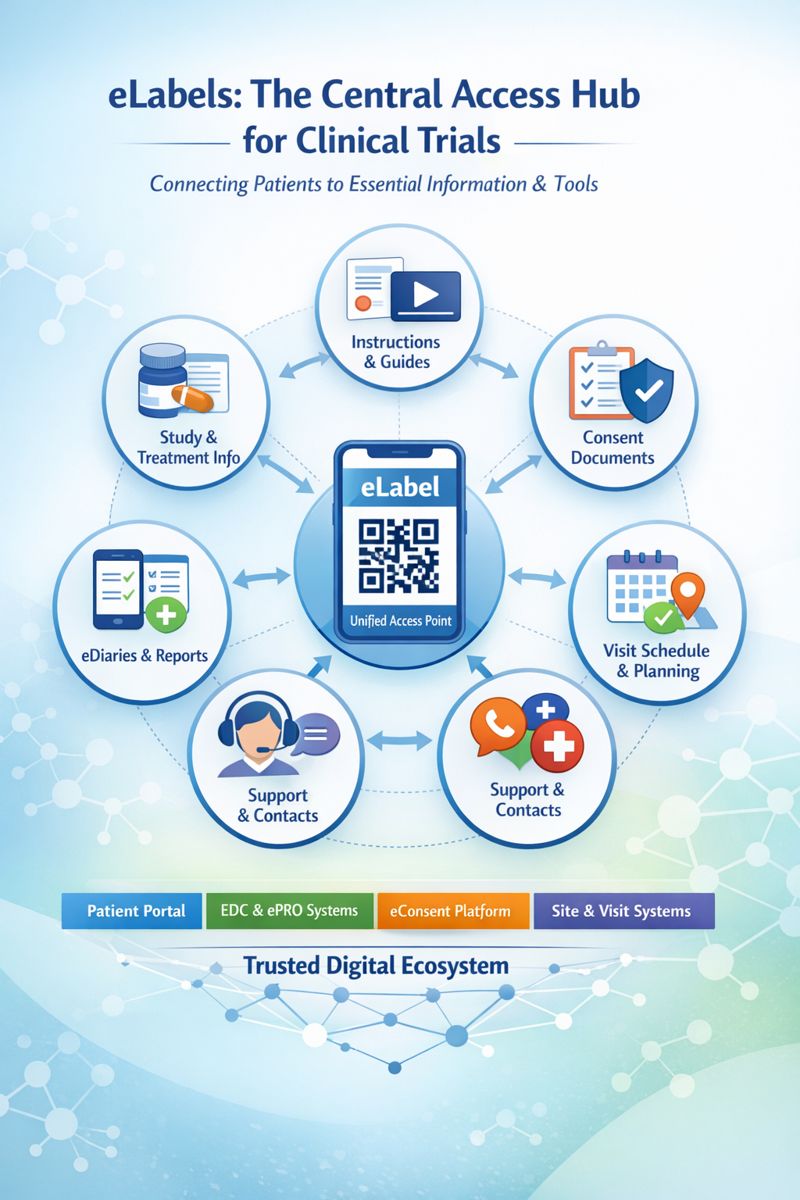
eLabels as the Natural Gateway to the Electronic Clinical Trial
Across a clinical trial, patients interact with many systems, documents, and sources of information—often at moments when clarity and simplicity matter most. Study details, instructions, digital tools, consent materials, visit schedules, and support channels all play a role, yet they are frequently fragmented across platforms. This fragmentation increases cognitive burden and can undermine the patient experience, even in well-designed trials.
eLabels offer a simple but powerful way to find it. By acting as a trusted, medication-centric access point, they connect patients to the information and systems they need, exactly when they need them. Rather than replacing existing solutions, eLabels complement them, providing a single, consistent gateway into the electronic clinical trial ecosystem. As trials continue to digitize, extensible eLabels are not just a convenience; they are a logical foundation for a more connected, patient-centered clinical trial experience.





